Functional genomic studies of Cdk8/CDK19 in development and disease
Almost 75% of genes responsible for human diseases are also found in the fruit fly Drosophila, which is genetically simpler, making it an ideal organism to study human disease mechanisms. We can engineer fruit flies to carry the same genetic mistakes found in human patients. We can then study their cell and organ functions and learn more about the causes of disease. We can use additional genetic tricks to find out if there are ways to reverse these disease features. We are studying equivalent genes called Cdk8 in flies and CDK19 in humans that are very important for ensuring that cells function properly. When this gene is defective in humans, it causes several developmental and neurological abnormalities. By generating fruit fly strains that are also lacking the equivalent gene, we can start to understand what happens in cells after this protein which is encoded by that gene is missing.
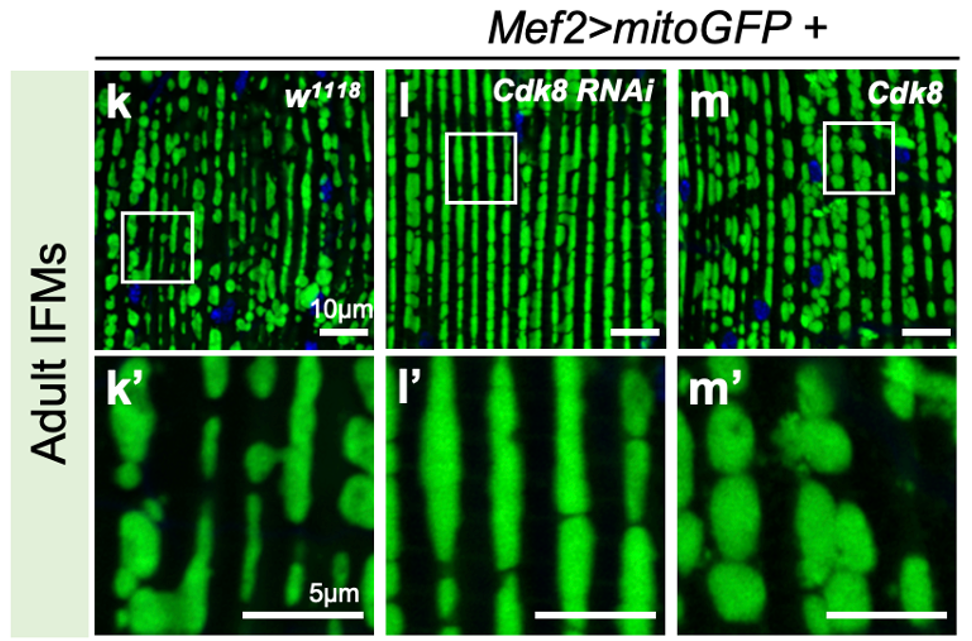
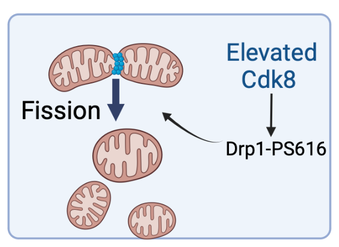
We found exciting new information that Cdk8 controls the shape and function of mitochondria. These organelles are essential for cell health and for generating energy inside our cells, and when they are defective many types of diseases can arise. We have been able to treat and cure mutant flies with the human CDK19 gene, which is a powerful way to show that these genes work the same way in both organisms. In addition to displaying features of the CDK8/19 disorders, we find that flies lacking Cdk8 have striking similarity to a Drosophila model of juvenile Parkinsonism caused by mutation of the Pink1gene. Pink1 is a kinase that regulates mitochondrial quality control, and disruption leads to impaired mobility and accumulation of abnormal mitochondria. We find that both Cdk8 and cytoplasmic CDK19 expression can rescue Pink1 behavioral defects in a new role for Cdk8/19 family members. This is exciting as it opens to areas to explore for therapeutics, using the knowledge we gain about Cdk8 and CDK19.
See our recent study published in Nature Communications.
This project is a collaboration with the labs of Dr. Hugo Bellen (Baylor College of Medicine) and Dr. Hyunglok Chung (Houston Methodist Research Institute). Funded by the Canadian Institutes of Health Research (CIHR).
We found exciting new information that Cdk8 controls the shape and function of mitochondria. These organelles are essential for cell health and for generating energy inside our cells, and when they are defective many types of diseases can arise. We have been able to treat and cure mutant flies with the human CDK19 gene, which is a powerful way to show that these genes work the same way in both organisms. In addition to displaying features of the CDK8/19 disorders, we find that flies lacking Cdk8 have striking similarity to a Drosophila model of juvenile Parkinsonism caused by mutation of the Pink1gene. Pink1 is a kinase that regulates mitochondrial quality control, and disruption leads to impaired mobility and accumulation of abnormal mitochondria. We find that both Cdk8 and cytoplasmic CDK19 expression can rescue Pink1 behavioral defects in a new role for Cdk8/19 family members. This is exciting as it opens to areas to explore for therapeutics, using the knowledge we gain about Cdk8 and CDK19.
See our recent study published in Nature Communications.
This project is a collaboration with the labs of Dr. Hugo Bellen (Baylor College of Medicine) and Dr. Hyunglok Chung (Houston Methodist Research Institute). Funded by the Canadian Institutes of Health Research (CIHR).
Studying cancer in Flies
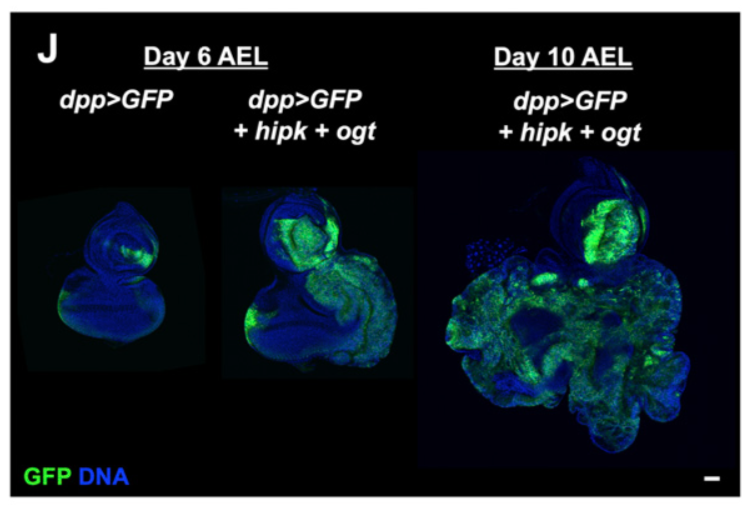
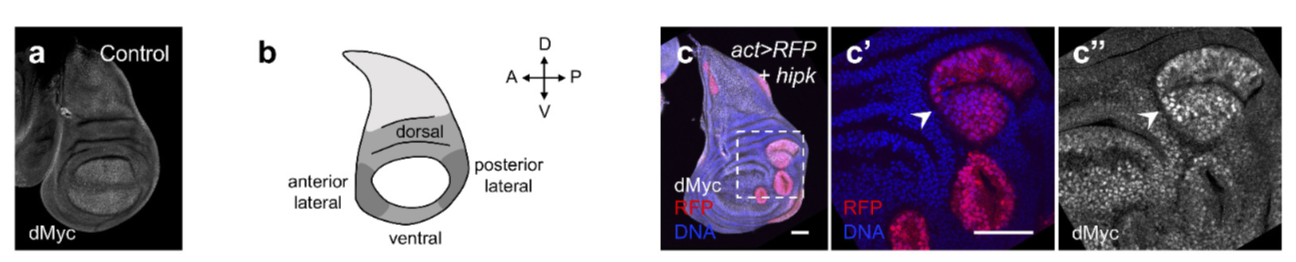
We have been studying “Hipk” proteins and their regulation of organ growth. Hipk proteins are found in many species including humans. We and others have found that loss of Hipk/Hipk2 in both mice and flies results in reduced proliferation, suggesting that Hipk family members are required for growth. We generated a novel cancer model in flies that is due to having too much Hipk protein. This causes fly organs and limbs to overgrow massively. In addition, cells bud off from these organs and move to new locations within the body, dramatically mimicking what occurs during metastasis of cancers in humans. We are currently trying to understand how Hipk hijacks the normal cellular control mechanisms to cause cells to become rogue and initiate tumors and metastasis. We hope to find ways to reverse the effects of Hipk on cells, leading to the therapies that can be used to treat certain cancers.
See our recent study published in Frontiers in Cell and Developmental Biology.
Funded by the Canadian Institutes of Health Research (CIHR).
See our recent study published in Frontiers in Cell and Developmental Biology.
Funded by the Canadian Institutes of Health Research (CIHR).
Control of organ formation and morphogenesis during development
The long-term goal of my research program is to understand how groups of cells develop into specialized adult organs. Such a process requires regulation of growth, morphogenesis and differentiation. My research group uses the well-characterized, genetically-tractable fruit fly, Drosophila melanogaster, as a model organism. Specifically, we seek to understand how developmental processes are regulated by protein kinases, as precise regulation of signal transduction pathways is often achieved through the reversible phosphorylation of proteins.
Funded by the Natural Sciences and Engineering Research Council of Canada (NSERC)
Modeling Robinow Syndrome in Drosophila and Chick
We are studying autosomal dominant Robinow syndrome (RS), in which genetic changes affecting WNT signaling pathways have been found. Major clinical features include shortening of the limbs and craniofacial abnormalities, suggesting fundamental defects in skeletal tissue organization. In collaboration with Dr. Joy Richman at UBC we are using the chicken embryo and fruit fly to study the links between genotype and phenotype. By expressing human gene variants in animal models, we learn not only about the impact of the mutation on gene function but also about how polarity in vertebrate cells contributes to morphogenesis.
Funded by the Canadian Institutes of Health Research (CIHR).
Funded by the Canadian Institutes of Health Research (CIHR).